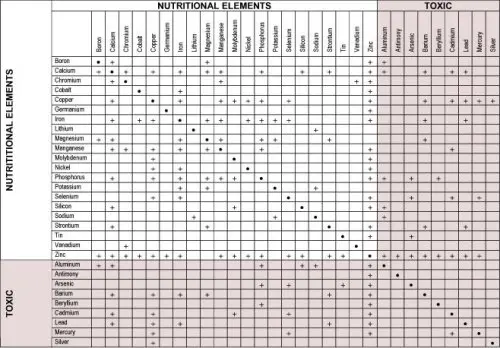
Tin Trace Mineral and Toxicity
Food absorbs tin from cans, so we ingest it, but it's poorly absorbed so unlikely to cause toxicity.
Tin was only recently identified as essential, but its precise function remains elusive. Scientists noted a 24% to 59% acceleration in growth when tin was supplied to baby rats on tin-deprived diets. This hints of some very important role for Tin and a possible relationship to growth hormone. Thus, Tin may be a player in our struggle to trade fat for muscle. Dietary intake of tin has been sharply reduced over the past 20 years. Supplementation is desirable.
There has been a tremendous amount of interest lately over the relationship of adult production of growth-hormone, and quality of life and longevity. It seems that the same hormone that helps kids grow, also helps adults defy their chronological age and take on youthful characteristics of a much younger biological age and level of cellular activity.
While most of the attention is focused upon the amino acid portion of the growth hormone equation (and while most of us get plenty of protein) it should be noted that young lab animals deprived of Tin failed to grow – so there may be a GHT in relationship. Also, as discussed above, Boron is useful for providing these same youthful hormone type benefits. And for serious longevity enthusiasts, it should be noted that Coenzyme Q-10 extended the lives of lab animals to the human equivalent of 150 years while greatly extending the quality of life to match that added time.
Tin or stannum in Latin, is essential in some mammals, including rats, but it has not been shown to be needed in humans. It is more often considered a mildly toxic mineral. One of the reasons its essentially is questionable in humans is its absence in newborns and in many animals. It is present in the earth in very small amounts. Tin is most often thought of as an environmental contaminant, both as tin cans and as inhaled industrial pollution. Levels, especially in the lungs, increase with age.
Primitive humans had much less tin in their bodies than modern humans. Tin has been used since the Bronze Age began more than 3,500 years ago (bronze contains copper and tin). It has been used for food storage for more than 200 years. Food does absorb tin from cans, and so we ingest this tin. Luckily for us, it is poorly absorbed from the gastrointestinal tract, probably less than 5 percent, so this is not likely to cause toxicity. Most excess tin is excreted in the feces. Some is eliminated in sweat and even less in urine.
Sources of Tin Toxicity
Tin is present in very low amounts in the soil and in foods. Canning, processing, and packaging often add some tin to food; the solder in iron or copper pipes contains tin; stannous fluoride in toothpaste may add more. Since we consider tin primarily as a contaminant, though fairly nontoxic, we should try to avoid it. Using few canned foods and avoiding toothpaste with tin are some ways to do this.
Functions
There are no known functions for tin in humans. If it has a function, it may be related to protein structure or oxidation and reduction reactions, though tin is generally a poor catalyst. Tin may interact with iron and copper, particularly in the gut, and so inhibit absorption.
Uses
No uses for tin are presently known other than in food storage, in industrial processes, and as a fluoride carrier in toothpaste.
Deficiency and Toxicity
Though tin is considered a mildly toxic mineral, there are no known chronic or serious diseases from tin exposure or ingestion. Studies in rats showed mainly a slightly shortened life span. I have found no cases of acute tin exposure; chronic low-level environmental and food contamination is more likely. Avoiding eating too much food from tin cans is probably the best we can do (oily foods seem to pick up more tin than others). In the United States, tin cans are now lacquered, which prevents some food absorption. Lacquered cans have a slight yellow coloring, while the unlacquered cans, which are more common with imported foods, are brighter metal. There are no known problems from tin deficiency in humans.
Requirements
There is currently no RDA or any known requirement for tin. We should avoid any large or long-term exposure. The average diet may contain about 2 mg. per day, but this can vary from about 2-20 mg. per day, depending on the foods ingested. Tin is not likely to be found in many supplements other than occasional trace mineral formulae, and there is no current reason to add tin to any nutritional program.
![[CRSN016] Tin](/web/image/product.template/3470/image_1920/%5BCRSN016%5D%20Tin?unique=88ee475)